Ionic compounds form when the atoms of a metal combine with the atoms of a non - metal
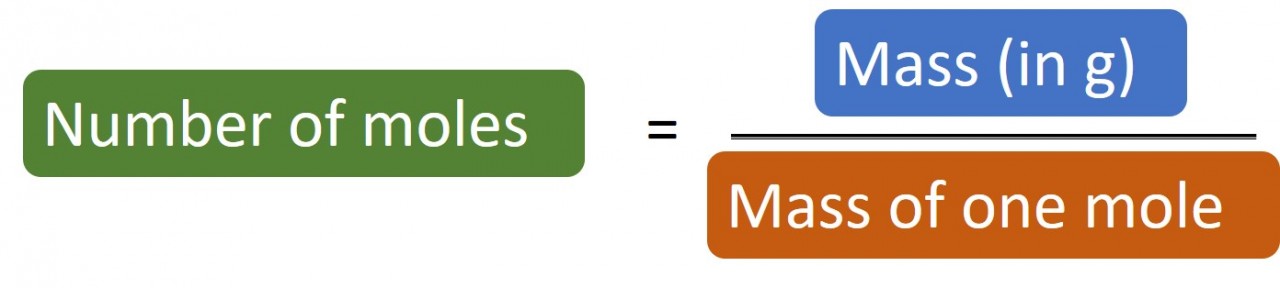
1.27 What is a mole? Students should: 1.27 know that the mole (mol) is the unit for the amount of a substance 1.28 understand how to carry...
1.28 Calculating moles The following masses of elements all contain one mole of atoms: 12.0 g Carbon, 32.1 g Sulphur, 14 g Nitrogen,&n...
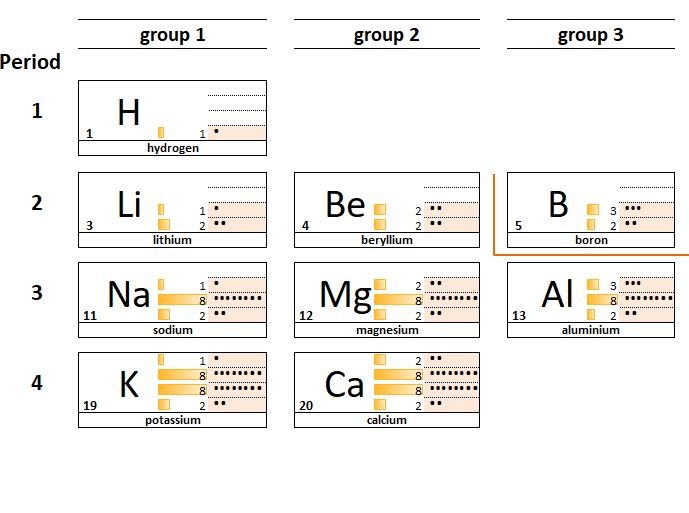
1.23 Family resemblances Hydrogen is often listed above group 1 the Alkali metals. Although like group 1 atoms, hydrogen atoms have a ...
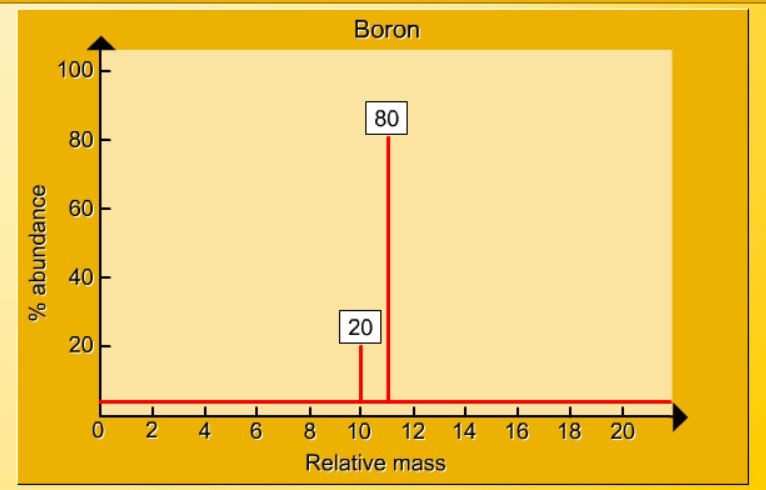
1.17 Activity 2. Calculating relative atomic mass. Students should: 1.17 be able to calculate the relative atomic mass of an element (Ar) ...
In the "furry worm" experiment the reactive magnesium metal reduces the less reactive copper. In this post we are going to investigate how...
Intro image Enter your text here ... Enter your text here ... Enter your text here ... Enter your text here ... Enter your text here ... Enter your te...