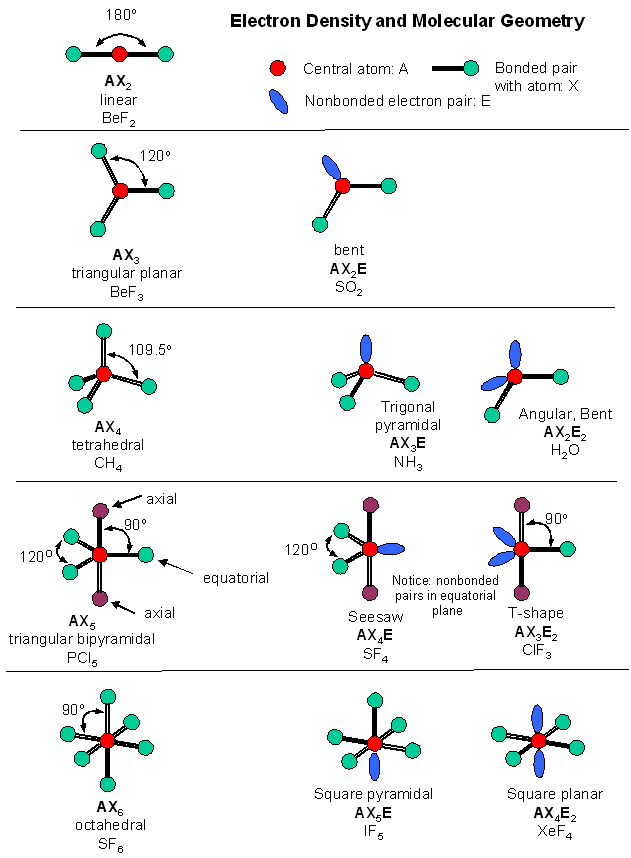
Methane, Ammonia and Water
Accounting for electrons.
The images in this slideshow here show how we can account for the way in which electrons are shared between atoms.
Use the first of the three buttons to the top left of the animation to see the outer electron configuration of each atom. Construct a table to show the name of the molecule, formula, no.of bonding pairs and number of non-bonding pairs:
Enter your text here ...
Predicting angles
Use the animation to find out the bond angle in the methane, ammonia and water molecules.shown
What is the connection between the number of non bonding pairs, bonding pairs and the bond angle?
Enter your text here ...
Enter your text here ...