Enter your text here ... The halogens are a family of elements in group 7 (aka 17) of the periodic table. in this video, you can see how reactive ...
Proton NMR produces a spectrum with a number of peaks. The number of peaks corresponds to the number of different chem...
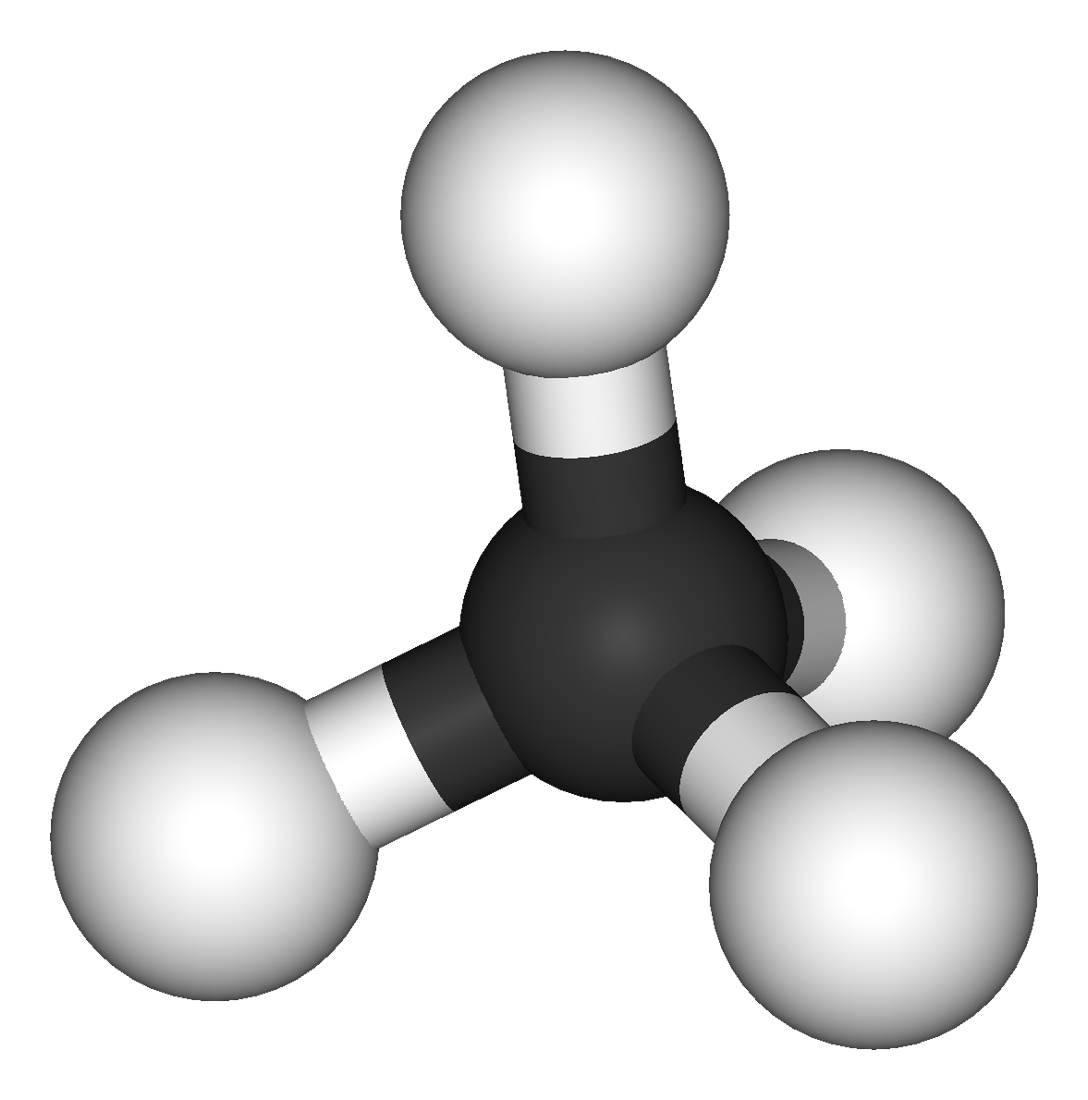
the alkanes are a family of hydrocarbons. hydrocarbons are compounds containing hydrogen and carbon only. methane is the simplest hydrocarbon wit...
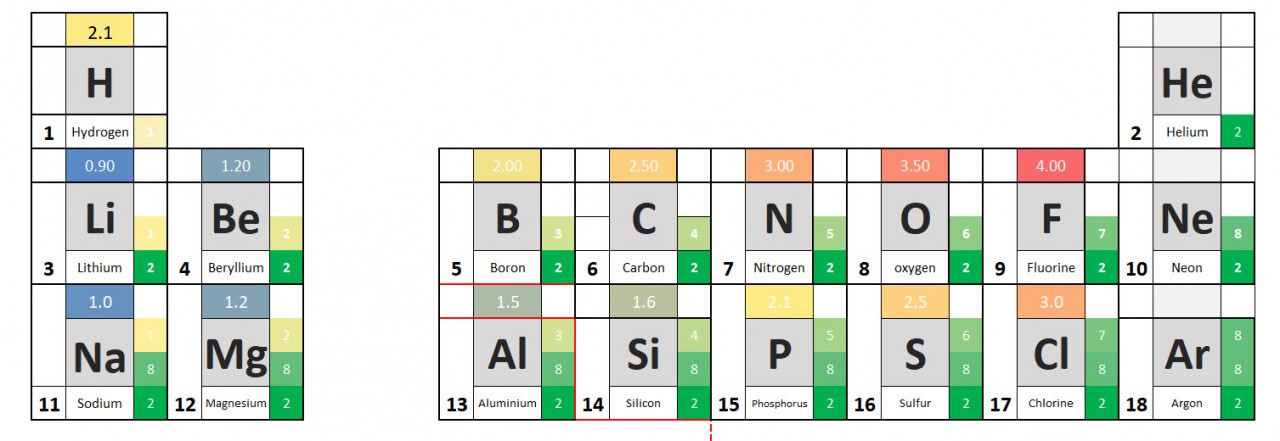
Electronegativity values allow us to predict the direction and strength of dipoles which might exist in molecules
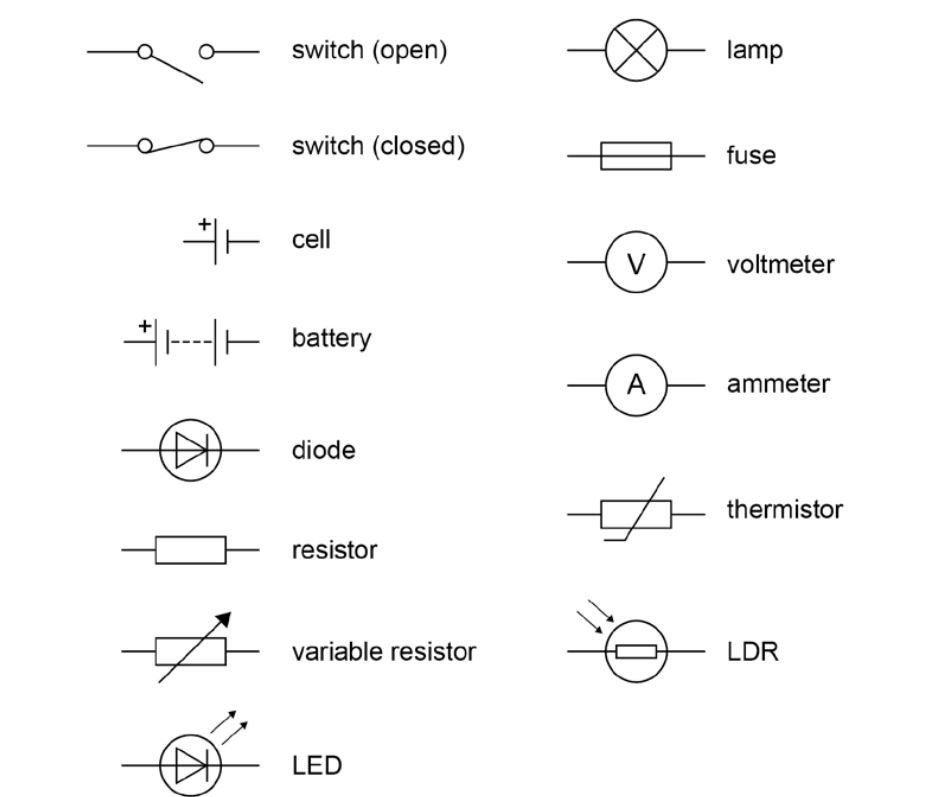
Symbols used in electrical circuit diagrams: Enter your text here ... Enter your text here ...
1.34 understand that ionic compounds have high melting and boiling points because of strong electrostatic forces between oppositely charged ions1.35 ...
Ionic compounds form when the atoms of a metal combine with the atoms of a non - metal
Ionic compounds form when the atoms of a metal combine with the atoms of a non - metal
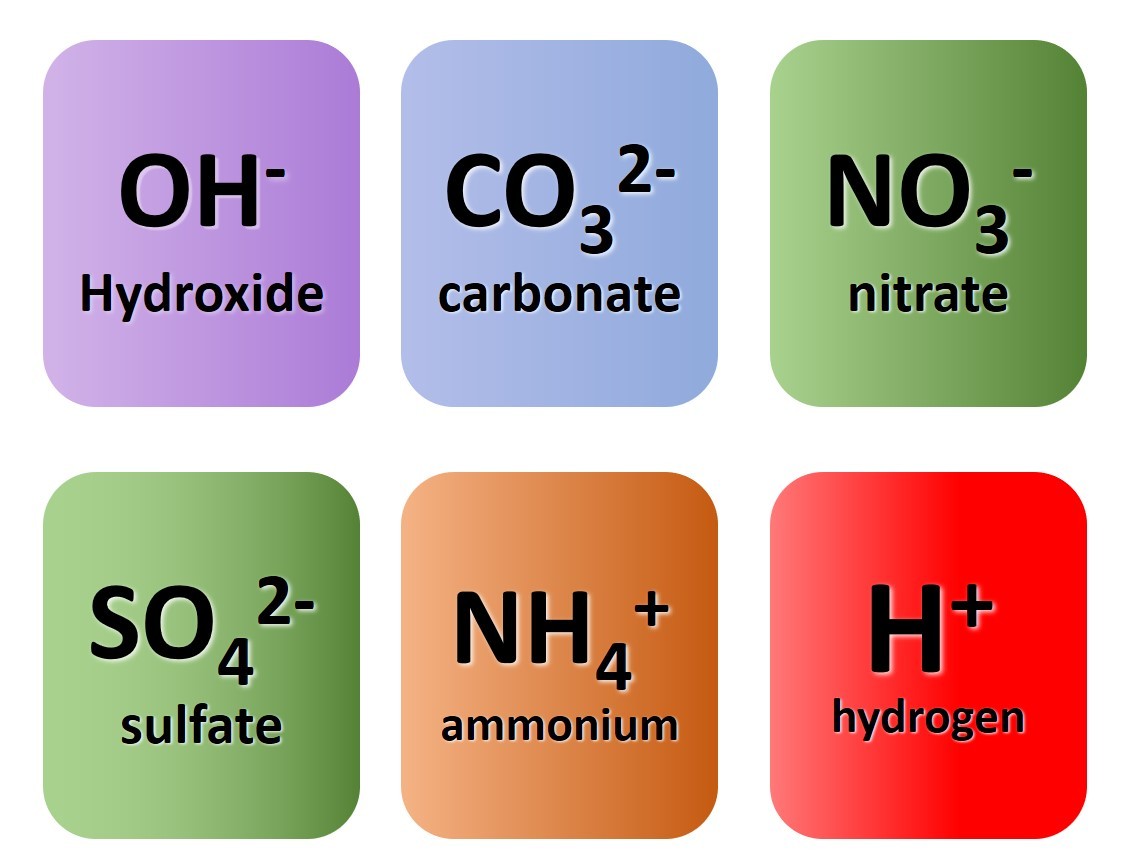
1.38 Activity. Know your ions. Students should: 1.38 know the charges of common ions listed You will have noted from the video that metal ...
1.39 Activity. Finding formulae Students should: 1.39 write formulae for compounds formed between the ions listed Watch the video closely ...
2.15 - 2.20 Loss and gain Oxidation and reduction reactions are very commonplace. When a element combines with oxygen that element is said to ha...
Before tackling this topic make sure you are able to: explain the difference between intermolecular and intramolecular forces. use...
1. Why is water a liquid and not a gas ( between 0 and 100oC) ? Water has a Relative Molecular mass ( Mr) of 18 ... Oxygen's Mr is 32. ...
Enter your text here ... Introduction Every chemical reaction has an accompanying change in energy. Some reactions release energy to the surroundings...
If a nucleus was the size of a raisin, the rest of the atom would be the size of a sports stadium
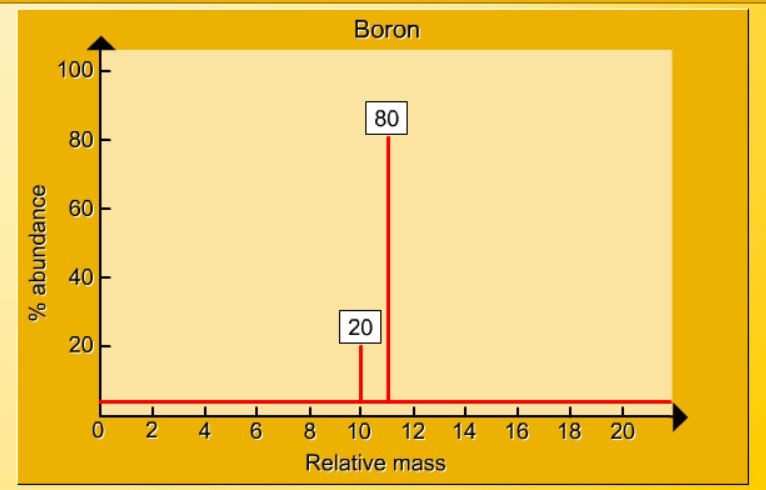
1.17 Activity 2. Calculating relative atomic mass. Students should: 1.17 be able to calculate the relative atomic mass of an element (Ar) ...
1.16 Activity 1. Three types of carbon atom Students should: 1.16 know what is meant by the terms atomic number, mass number, isotopes and...
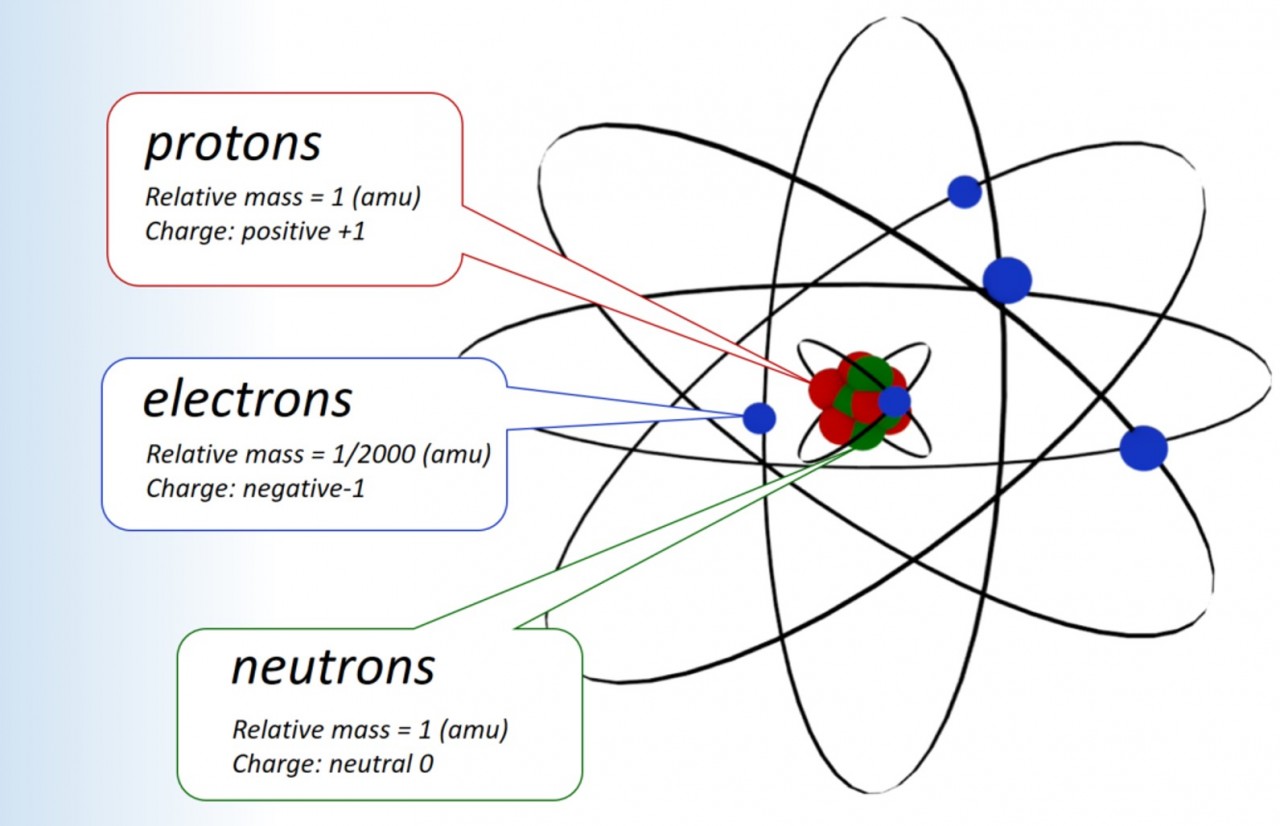
1.15 Sub atomic particles Students should: 1.15 know the structure of an atom in terms of the positions, relative masses and relativecharg...
If a nucleus was the size of a raisin, the rest of the atom would be the size of a sports stadium
Atomic models Atoms are the building blocks of matter. Atoms are the smallest possible particles into which an element can be subdivided, withou...